New atlas of proteins could reveal the cause of dementia and various cancers
Scientists have developed a comprehensive atlas of proteins that reveals how these molecules behave within human cells.
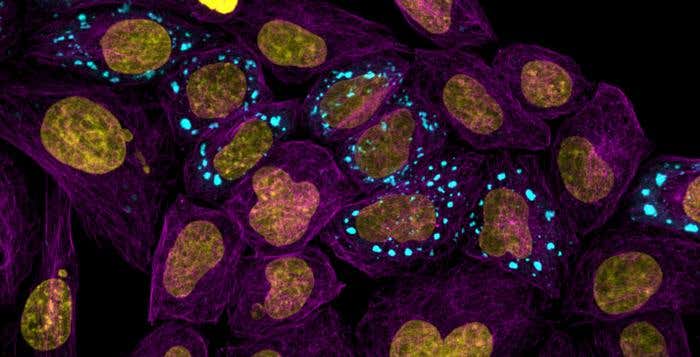
New groundbreaking work from scientists at the University of Cambridge has developed a comprehensive atlas of proteins that reveals how these molecules behave within human cells. This innovative tool has the potential to uncover the origins of protein-related diseases such as dementia and various cancers.
Published in Nature Communications, this atlas allows researchers to pinpoint new proteins within cells responsible for crucial bodily functions. The team behind this discovery focuses on a droplet-like cell structure known as a condensate. These condensates serve as organizational hubs where proteins gather and are also critical sites where disease processes can begin.
What makes this atlas so valuable is its accessibility; researchers worldwide can use it to explore their protein targets of interest and the associated condensate systems.
“This model has allowed us to discover new components in membrane-less compartments in biology as well as discover new principles underlying their function,” explained Professor Tuomas Knowles, who led the research.
Cells maintain their structure and function through a meticulous organization of molecules, one of which involves gathering inside condensates. These microscopic hubs are essential for the machinery that powers living cells. Until now, scientists lacked a comprehensive map detailing which proteins converge within which condensates. Knowles highlighted, “To date, we have not had a comprehensive map of which proteins go together into which condensates, but in our work, we provide a first such atlas.”
The behavior of proteins inside cells remains partially understood, prompting the team to create this atlas to predict protein interactions within condensates. “What motivated this research was the desire to understand the full complexity of protein condensates and to go a layer deeper than what scientists have researched so far,” said Dr. Kadi Liis Saar, the first author of the research and a postdoctoral fellow at the Centre for Misfolding Diseases.
Related Stories
To achieve this, the researchers leveraged extensive databases like StringDB and BioGRID, which hold data on various cellular aspects, along with detailed case studies on individual condensates. The power of artificial intelligence (AI) allowed them to integrate this vast, complex, and often incomparable data. While previous research focused on a limited number of proteins, this atlas can characterize the entire cellular landscape.
“With this atlas, we can make predictions about every single protein in a cell, where exactly it would be found, and what sorts of other proteins it interacts with,” Saar commented. “We hope that this generates opportunities for researchers and opens up new possibilities for intervention in diseases associated with aberrant condensate formation.”
The AI-driven approach also led to the discovery of proteins within the model cell that had never been observed before. These findings, once verified in the lab, could confirm the AI's accuracy. “In our study, we discovered proteins within condensates that have never been seen there before.
These proteins are involved in important functions in the body, such as the distribution of fat, the creation of actin inside cells, and the creation of new proteins,” noted the researchers. These proteins were absent in the previous study that served as the training set for the AI model.
“We hope these data will enable new discoveries about the biological roles of condensates as well as the biophysical drivers behind condensate formation,” the team added.
This atlas represents a significant advancement in understanding cellular behavior and protein interactions. It holds promise not only for basic biological research but also for developing new treatments for diseases linked to protein misbehavior. The combination of large-scale data integration and AI has opened a new frontier in cell biology, paving the way for discoveries that could transform our approach to diagnosing and treating complex diseases.
Note: Materials provided above by the The Brighter Side of News. Content may be edited for style and length.
Like these kind of feel good stories? Get the Brighter Side of News' newsletter.
Joshua Shavit
Science & Technology Writer | AI and Robotics Reporter
Joshua Shavit is a Los Angeles-based science and technology writer with a passion for exploring the breakthroughs shaping the future. As a contributor to The Brighter Side of News, he focuses on positive and transformative advancements in AI, technology, physics, engineering, robotics and space science. Joshua is currently working towards a Bachelor of Science in Business Administration at the University of California, Berkeley. He combines his academic background with a talent for storytelling, making complex scientific discoveries engaging and accessible. His work highlights the innovators behind the ideas, bringing readers closer to the people driving progress.